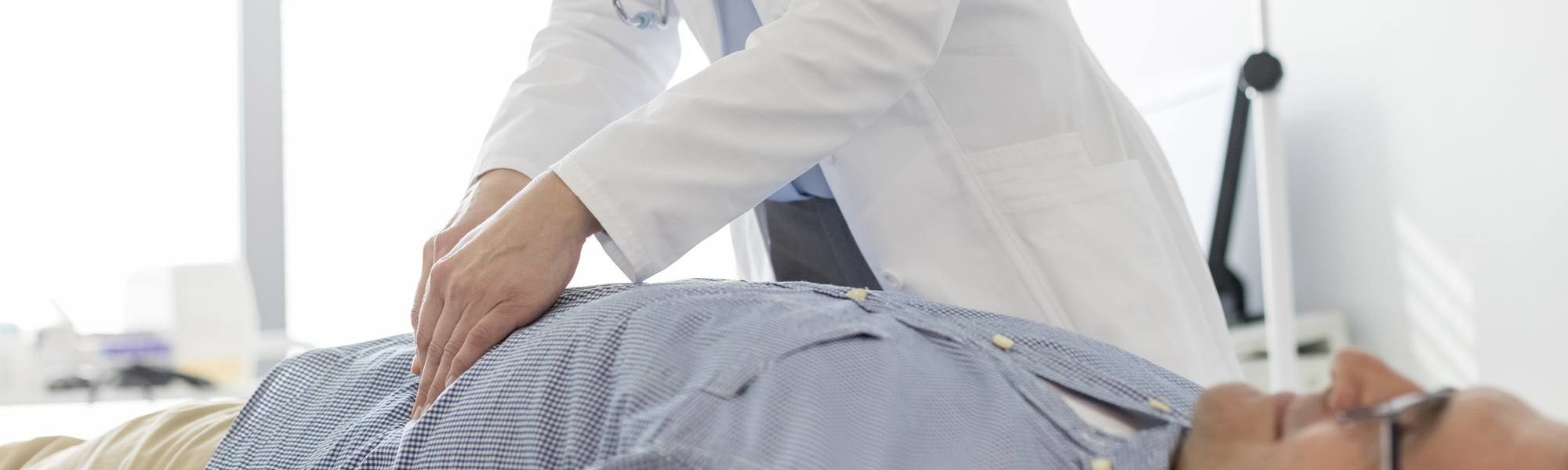
Gastroenterology clinical trials
Intense competition for patients, larger clinical trials, and for some indications, the need to identify and enroll patients in flare, make patient recruitment challenging for companies running gastroenterology clinical trials today.
AES has in-depth gastroenterology experience, including significant expertise in non-alcoholic steatohepatitis (NASH), non-alcoholic fatty liver disease (NAFLD), Crohn’s disease, ulcerative colitis and more.
Why AES for gastroenterology clinical trials?
AES brings the precise mix of expertise, insights, specialists and infrastructure necessary to overcome these formidable challenges. And we have the track record to substantiate our success.
AES Experience in Gastroenterology
A focus on Nonalcoholic Steatohepatitis (NASH)
The traditional model of relying on sites to find patients – which is ill-equipped to meet the needs of most trials – is particularly ineffective for nonalcoholic steatohepatitis NASH studies, in which the demand to recruit a largely asymptomatic and undiagnosed patient population is exploding.
Because relatively few NASH patients are referred to specialists, and fewer still will agree to undergo an invasive liver biopsy, only small, depleted databases of NASH patients are available at specialist sites. That’s why NASH study recruitment requires going beyond existing diagnosed patients and engaging with undiagnosed individuals with NASH comorbid conditions. But finding and pre-screening undiagnosed patients at scale is not something that most specialist sites can do. Our AES pre-screening and scheduling platform, however, has the capacity to evaluate 10,000 patients every day.
At the heart of our NASH recruitment solution is a database that allows us to mine vast pools of undiagnosed individuals whose comorbid conditions place them at a high risk of NASH (63 million over age 50, 57 million with obesity, 21 million with hypertension and 10 million with diabetes).
Our pre-screening process – which includes FibroScan® and liver function tests at one of our NASH-enabled AES sites – identifies subjects with probable NASH. Those subjects who meet all eligibility criteria can receive biopsy counseling and a referral for the more invasive screening procedure.
Learn more about our NASH experience in this video.
A focus on Inflammatory bowel disease (IBD)
IBD is a disease with significant unmet medical needs. Through global surveys of more than 800 Crohn’s and ulcerative colitis patients, AES found that IBD patients are frustrated by inadequate symptom relief from current treatments, the need to take multiple medications, and the ongoing search for new treatment options to manage their symptoms. In addition, patients are looking for greater control over the disease and freedom from the impact of IBD in three primary areas:
In the past five years, AES has randomized more than 400 IBD patients across seven studies, executed IBD trials at 43 AES sites in 14 countries, and increased enrollment rates for IBD trials at non-AES sites by up to 60%.
Track your spectrum of IBD symptoms
Despite the need for new treatments, IBD trials have low and declining enrollment rates. In Crohn’s disease trials, over the last 10 years, the patient enrollment rate has declined by 50%, and for ulcerative colitis trials, over the last five years, the patient enrollment rate has declined by 13%.1
To counter declining enrollment rates, companies conducting IBD trials – like those across most indications – typically add research sites, thereby increasing costs, change orders and timelines. For example, over the last 10 years, the average number of research sites in Crohn’s disease studies has increased by 193%.1
Our approach to enrolling IBD patients is targeted and proven. We leverage a database of more than 18 million potential gastrointestinal patients – including 1.8 million people self-reporting Crohn’s disease or ulcerative colitis – and augment our recruitment efforts with referrals from our network of gastrointestinal specialist partners throughout the world. Then, to solve one of the biggest challenges of recruiting patients for IBD trials – finding and enrolling patients in flare – we regularly re-engage with pre-qualified patients using our MyColo mobile app and telephone outreach. In this way, we are able to identify patients in flare and quickly schedule them at their local clinical trial site in time to enroll in the study.
In addition, if you choose our Integrated Network & Patient Recruitment solution, which recruits volunteers and patients directly to AES-owned research sites, we can also provide more than 40 dedicated research sites with teams that have previously run IBD clinical trials.
Sources
- GlobalData, AES analysis. March 2021.

